
By Nitin Yadav, Researcher, NITISARA
This article talks about the availability and strategic sourcing of medical devices, including the opportunity for global supplier discovery and procurement market share. Further, we discuss expected growth, patterns of imports and exports and important government programs, international partnerships, investment patterns, and the structural problems that affect production in the home country.
Introduction
The medical devices industry in India is growing quickly. It is USD 20.51 billion by 2029 with a CAGR of 5.35%. India is one of the best places to do business in Asia, but it still gets 70% to 80% of its medical devices from other countries. This means that there is a lot of room for new ideas and making things in the area. To fill this gap, the government has started programs like the PLI schemes and the National Medical Devices Policy 2023. The goal of these programs is to get more money and make more things in the country. India wants to become a global MedTech hub by 2029, when it wants to export $20.51 billion worth of goods and improve healthcare in rural and digital areas. Over 70% of high-end equipment production is concentrated in the United States, Germany, Japan, and China — creating dependency risks for developing economies. The pandemic-driven surge in demand for ventilators, PPE kits, and diagnostic systems saw equipment prices spike by up to 200%, highlighting the need for diversified sourcing, strategic reserves, and transparent procurement mechanisms. Consequently, international agencies and governments are increasingly adopting resilient procurement models, including framework agreements, local manufacturing tie-ups, and digital tender platforms, to ensure continuity in access to critical medical technologies and enhance healthcare preparedness globally.
Global trade analysis
From its estimated USD 15.35 billion in 2023, the Indian medical device market is projected to grow at a compound annual growth rate (CAGR) of 5.35% to reach USD 20.51 billion by 2029. India ranks fourth in Asia, behind China, Japan, and South Korea, with 1.65% of the global market. In the Union Budget 2025-26, Rs. 99,858 crore (US$11.48 billion) was allocated as a budget for the healthcare sector. India and Russia have set the bilateral trade target at Rs. 260,880 crore (US$30 billion) by 2025. Trade is expected to increase by an additional Rs. 43,480 crore (US$5 billion) per annum, with opportunities in pharmaceuticals & medical devices, minerals, steel, and chemicals.
The sector is divided into Electronic equipment – 56%, Disposables & consumables – 26.5%, In-vitro diagnostics – 8.1%, Implants – 7.1%, Surgical instruments – 2.3%
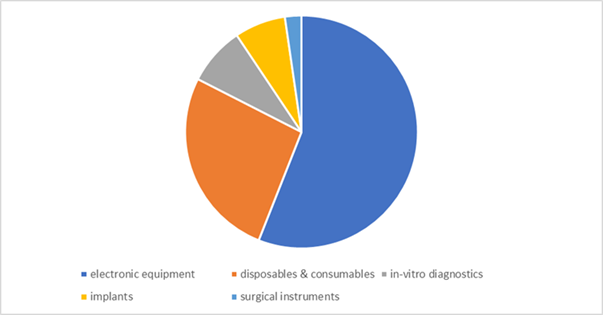
Import-Export Dynamics
With 70–80% of its medical device needs coming from overseas, India still depends largely on imports, indicating a chronic supply and demand imbalance. The Indian medical devices market is expected to increase from its estimated USD 15.35 billion in 2023 to USD 20.51 billion by 2029 at a compound annual growth rate (CAGR) of 5.35%. India currently has a 1.65% market share in the world market for medical devices.
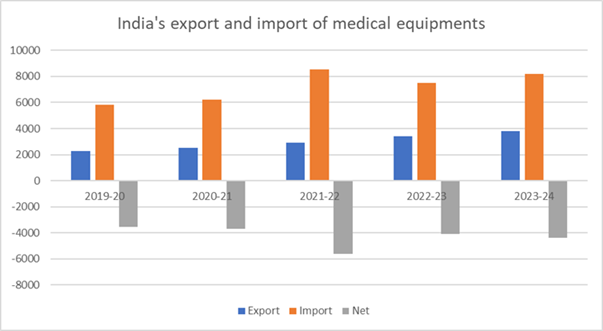
Medical device imports increased from Rs. 61,179 crore in FY 2023–2024 to Rs. 68,885 crore in terms of trade. Despite a minor decrease in 2022–2023, imports have risen by over 65% from Rs. 41,709 crore in 2019–20. Of all device categories, disposables saw the largest increase in imports, rising 17.63% to Rs. 4,090 crore, while imports of electronic equipment increased 14.5% to Rs. 44,132 crore. IVD reagent imports rose by 11%, consumables by 8.37%, and implants by 7%. The rise in imports is indicative of the country’s ongoing reliance on foreign medical technologies, even in the face of initiatives to increase domestic production.
Importing medical goods
The US, China, Germany, the Netherlands, and Japan send India most of its medical devices. These countries account for 66.6% of all medical device imports, which is ₹45,910 crore out of a total of ₹68,880 crore for the fiscal year 2023. The US has the most, with 22%. China comes next with 19%, Germany with 13%, the Netherlands with 7%, and Japan with 5%. The main reason these countries are so powerful is that they are home to some of the biggest and most famous multinational companies that make high-end, cutting-edge medical products. They are the best suppliers for India’s growing healthcare needs because they have a long history of making high-quality, new products that follow all the rules.
| Country | Company | Key Product Lines / Focus Areas |
|---|---|---|
| United States | Medtronic | Cardiac implants, neurostimulation devices, insulin pumps |
| | Abbott Laboratories | In-vitro diagnostics, diabetes management systems |
| | Stryker Corporation | Orthopaedic implants, surgical instruments |
| | GE HealthCare | Imaging systems (MRI, CT), patient monitoring, digital health |
| China | Mindray Medical | Patient monitoring, ultrasound, in-vitro diagnostics (IVD) equipment |
| | MicroPort Scientific | Cardiovascular implants, orthopaedic and neuro devices |
| | Lepu Medical | Stents, ECG machines, surgical devices |
| Germany | Siemens Healthineers | Advanced imaging (MRI, CT, X-ray), diagnostics |
| | B. Braun Melsungen | Infusion therapy, surgical instruments, hospital supplies |
| | Fresenius Medical Care | Dialysis machines and related consumables |
| Netherlands | Philips Healthcare | Diagnostic imaging, patient monitoring, connected care systems |
| | Medtronic(Regional Hub) | European manufacturing and logistics operations |
| Japan | Canon Medical Systems | MRI, CT, and ultrasound systems |
| | Terumo Corporation | Cardiovascular and endovascular systems, infusion technology |
| | Olympus Corporation | Endoscopy equipment, surgical visualization tools |
The Import License Process in India
Devices are classified based on risk levels into four categories: Class A (low risk), Class B (low-moderate risk), Class C (moderate-high risk), and Class D (high risk).
- Appoint an Indian Authorized Agent: Foreign manufacturers intending to import medical devices must appoint an Indian entity to act as their Authorized Agent. This entity should hold a valid wholesale or manufacturing license.
- Obtain Form MD-42: Before applying for an import license, the foreign manufacturer must ensure that their Authorized Agent has obtained a Medical Device Registration Certificate (Form MD-42) from CDSCO. This license authorizes the Indian Agent to import, sell, stock, exhibit, or distribute medical devices, including in vitro diagnostic devices, to retailers, wholesalers.
- Apply for Import License: The first formal step in the licensing process is the submission of Form MD-14. This application form is submitted to the CDSCO via the SUGAM portal, requesting approval to import specific medical devices into India.
- CDSCO Review and Grant of Form MD-15: After reviewing the Form MD-14 application and all supporting documents, the CDSCO will grant the Import License via Form MD-15 if everything is in order. The Form MD-15 Import License allows the importer (or their agent) to legally bring the medical devices into India and sell them within the Indian market.
- Customs Clearance & Post-Market Surveillance: Once Form MD-15 is issued, importers can proceed with customs clearance for their medical devices. However, compliance doesn’t end here. Importers must ensure ongoing adherence to India’s post-market surveillance requirements, including reporting adverse events and periodic safety updates to the CDSCO.
Government Support & Policies for domestic manufacturing
The CSIR Mission Mode Program aims to establish India as a leader in the manufacturing of high-end medical devices, with a focus on four key areas: advanced orthopaedic and dental implants, imaging-based medical applications, rehabilitation technologies, and diagnostic and therapeutic devices. Although most high-tech devices are currently imported, strong R&D and government support are driving efforts to reduce reliance on imports. The National Medical Devices Policy 2023, which intends to raise from $11 billion to $50 billion by 2030, and the Production Linked Incentive (PLI) scheme, which has approved 26 projects with investments exceeding Rs. 1,200 crore, are significant initiatives. The government has authorized medical device parks in states like Tamil Nadu and Himachal Pradesh to ensure quality and safety. The Indian Certification of Medical Devices (ICMED) program, budgetary allotments for healthcare and pharmaceuticals exceeding Rs. 86,000 crore, and the easing of import restrictions for necessary devices are additional sources of support. The goals of these initiatives are to increase domestic production, encourage innovation, draw in foreign capital, and establish India as a major hub for the production of medical devices.
Understanding the Factors Driving India’s Medical Device Imports
India’s massive reliance on imports for its medical device supply can be detrimental to its future supply chain. With almost 70-80 % of our demand fulfilled by foreign countries, India’s domestic manufacturing is suffering from its heavy competition as they are not able to fight back high-end supply of goods. A few of the reasons for this can be:-
- An adverse duty structure: Many medical devices manufactured overseas are less expensive than those made domestically because of the import duty system. This price advantage discourages investment in local manufacturing facilities.
- Inadequate Demand for Specific Products; Some specialized or cutting-edge medical devices are not very needed in India. Because of the small market size, local production is not economically viable for manufacturers.
- Inadequate Intellectual Property Protection: India lacks comprehensive laws and strong enforcement to protect the intellectual property of medical devices. As a result, companies are less inclined to produce locally and innovate.
- Lack of an Indigenous Quality Certification Authority; The absence of a recognized Indian certification body for medical devices affects consumer confidence in homegrown goods. Without local quality certification, Indian devices struggle to compete on a global scale.
- Inadequate Ecosystem Support; Manufacturing requires a robust supply chain, but many necessary components are still imported. The lack of a local ecosystem increases expenses and complicates production processes.
- A Complex Regulatory Environment: Delays and complexity are increased by the need for multiple regulatory approvals. These barriers make it difficult for producers to quickly launch goods into the Indian market.
- Lack of skilled workers and restrictive labour laws: There is a shortage of experts with the training required to produce cutting-edge devices. Additionally, rigid labor laws limit flexibility, which raises the cost of manufacturing.
In conclusion, the primary objectives of investing in India’s medical device sector should be to boost domestic manufacturing, advance R&D capabilities, and benefit from government initiatives such as PLI and medical device parks. Future prospects are presented by the development of state-of-the-art diagnostic tools, digital health technologies, and rural healthcare solutions. By 2030, building a strong, self-sufficient ecosystem and making India a global MedTech hub will necessitate international and local strategic partnerships in addition to investments in infrastructure for certification and skill development.
The views expressed do not represent the company’s position on the matter. Stay informed through the Nitisara Platform and Blogs, and adapt to emerging trends that are poised to thrive in the competitive global marketplace.- https://nitisara.org/category/blogs-updates/
References
- For precise details, visit https://cdsco.gov.in/opencms/resources/UploadCDSCOWeb/2018/UploadImmunization/11GUIDELINES1.pdf.
- https://tractus-asia.com/blog/indias-medical-device-sector/
- https://www.fortuneindia.com/macro/medical-devices-imports-from-china-us-up-41-in-fy22/108967
- https://www.business-standard.com/industry/news/imports-of-medical-devices-rise-21-till-oct-on-the-back-of-rising-imports-124011600105_1.html
- https://www.trade.gov/market-intelligence/india-medical-devices
- https://trendeconomy.com/data/h2/India/9018
- https://journals.lww.com/ijph/fulltext/2024/04000/covid_19_and_the_medical_devices_in_india__trade.14.aspx
- https://www.moneycontrol.com/news/business/indias-dependence-on-imported-medical-devices-at-alarming-level-industry-group-says-8890141.html









